Medical Device Manufacturing Specialist Directory Website Template
Assay is a single-column flow landing page built for medical device quality testing laboratories. It combines a full-screen video header, an industry-report content rhythm, and a gated PDF conversion path to serve regulatory affairs directors, quality engineers, and contract manufacturers who need to communicate rigorous testing credentials with authority and clarity.
by Rocket studio
Quick summary
Assay is a precision landing page template for medical device quality testing labs. It guides technically sophisticated visitors through an evidence-layered scroll experience, from a cinematic lab video header down through capability matrices, anonymized case-study tables, and a gated testing roadmap download. The design and content rhythm are built to establish trust before asking for anything.
Who this template is for
This template is designed for specialized testing and verification labs that serve the medical device industry. The content structure and conversion path are calibrated for audiences with regulatory and procurement responsibilities.
- Regulatory affairs directors managing 510(k) submission timelines who need to quickly verify a lab's testing scope
- Quality engineers at orthopedic or implantable-device startups requiring biocompatibility panel documentation aligned to ISO 10993
- Contract manufacturers whose original equipment manufacturer customers demand third-party verification before releasing purchase orders
What problem this template solves
Most laboratory websites describe their capabilities in general terms. Visitors with regulatory deadlines need specific, structured evidence before they can act. This template solves the credibility gap between claiming expertise and proving it.
- Visitors can assess testing scope through organized capability matrices before filling in any form
- The ungated data tables and regulatory-timeline infographic give away real value early, reducing friction and building confidence
- The gated PDF conversion path captures qualified leads without interrupting the evidence-building scroll experience
What you get with this template
You get a fully structured single-column flow landing page built around a deliberate text-then-data rhythm. Every section is designed to function like a chapter in a formal industry report.
- A full-screen video background header with an evergreen headline overlay and a semi-transparent loam overlay treatment
- Two ungated data tables and a regulatory-timeline infographic embedded in the scroll flow before any form appears
- A primary gated download form for the Testing Roadmap PDF and a secondary persistent call-to-action button for test plan review requests
Feature list
This section details the core built-in capabilities of the Assay template as defined by the source brief.
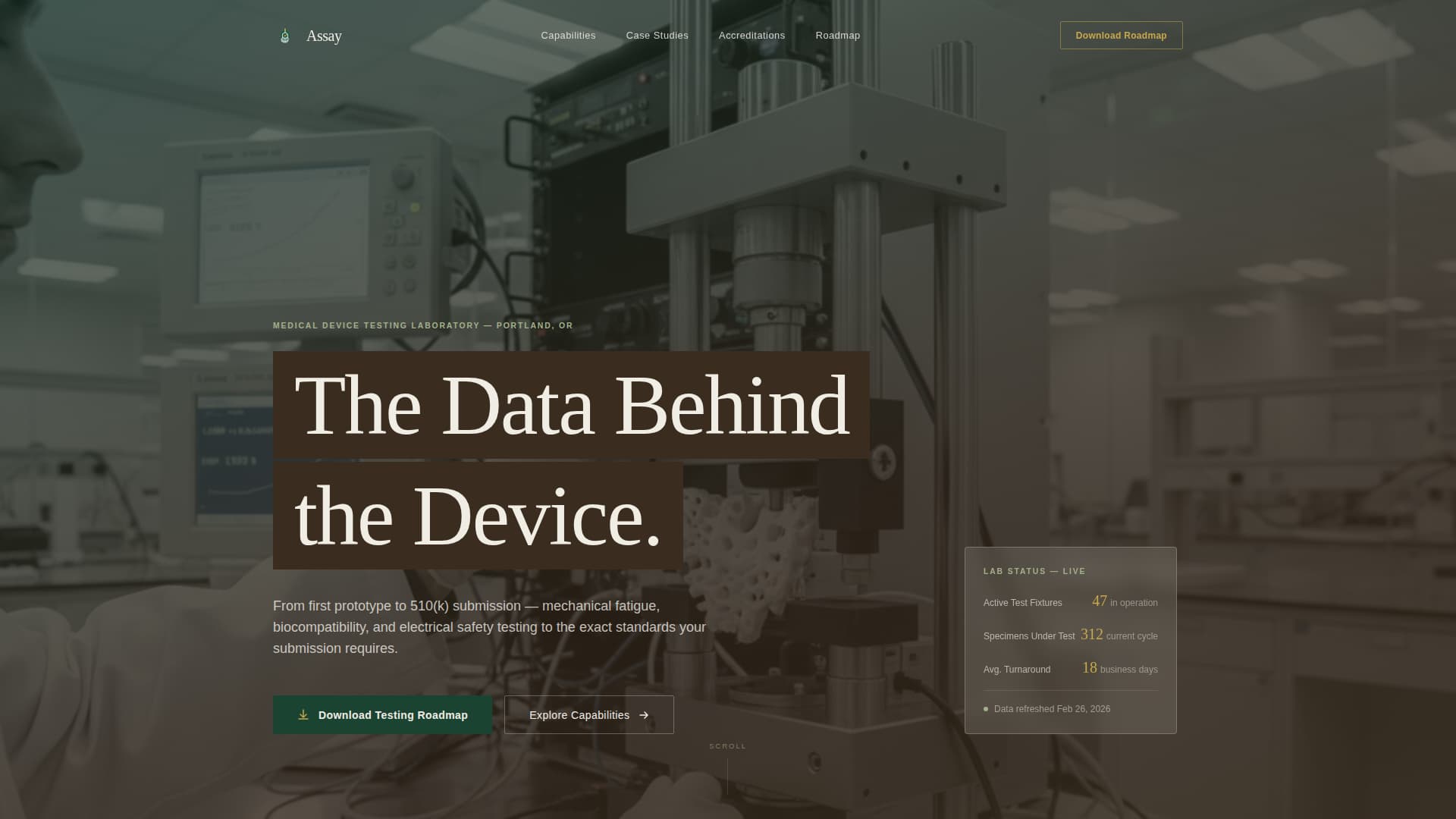
Full-Screen Video Background Header
The header occupies the full viewport with a slow, stabilized tracking shot moving through the lab at waist height. The footage is color-graded cool and desaturated. The headline "The Data Behind the Device" appears in birch-bark white over a semi-transparent deep loam overlay, letting typography emerge without competing with the footage.
Industry Report Content Rhythm
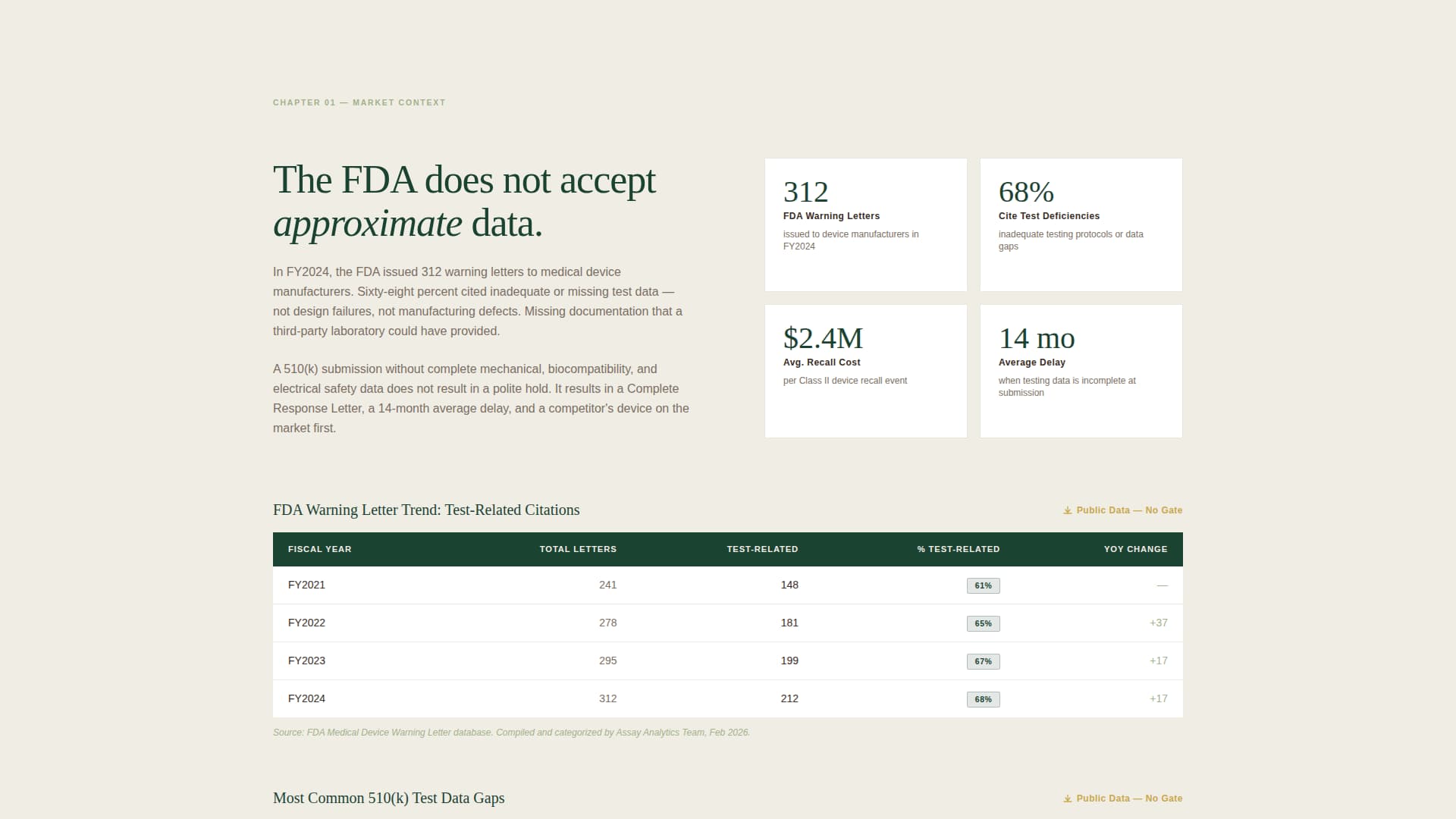
Each scroll section follows a deliberate text block, data visual, text block, data visual cadence. This mirrors the structure of a formal white paper. The sequence moves visitors from market-context paragraphs citing FDA warning-letter statistics into a capability matrix organized by test standard, then into anonymized case studies presented as data tables.
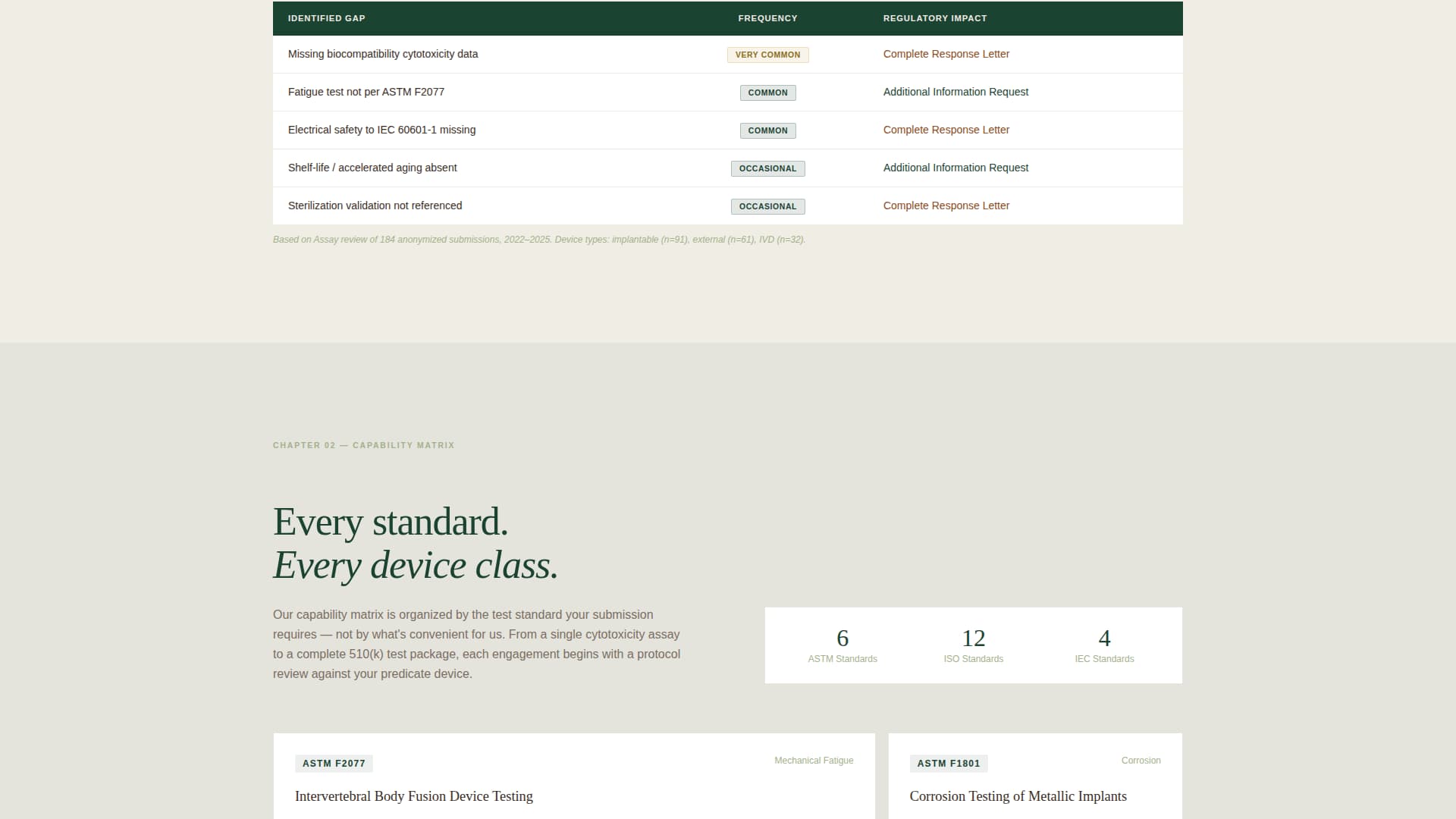
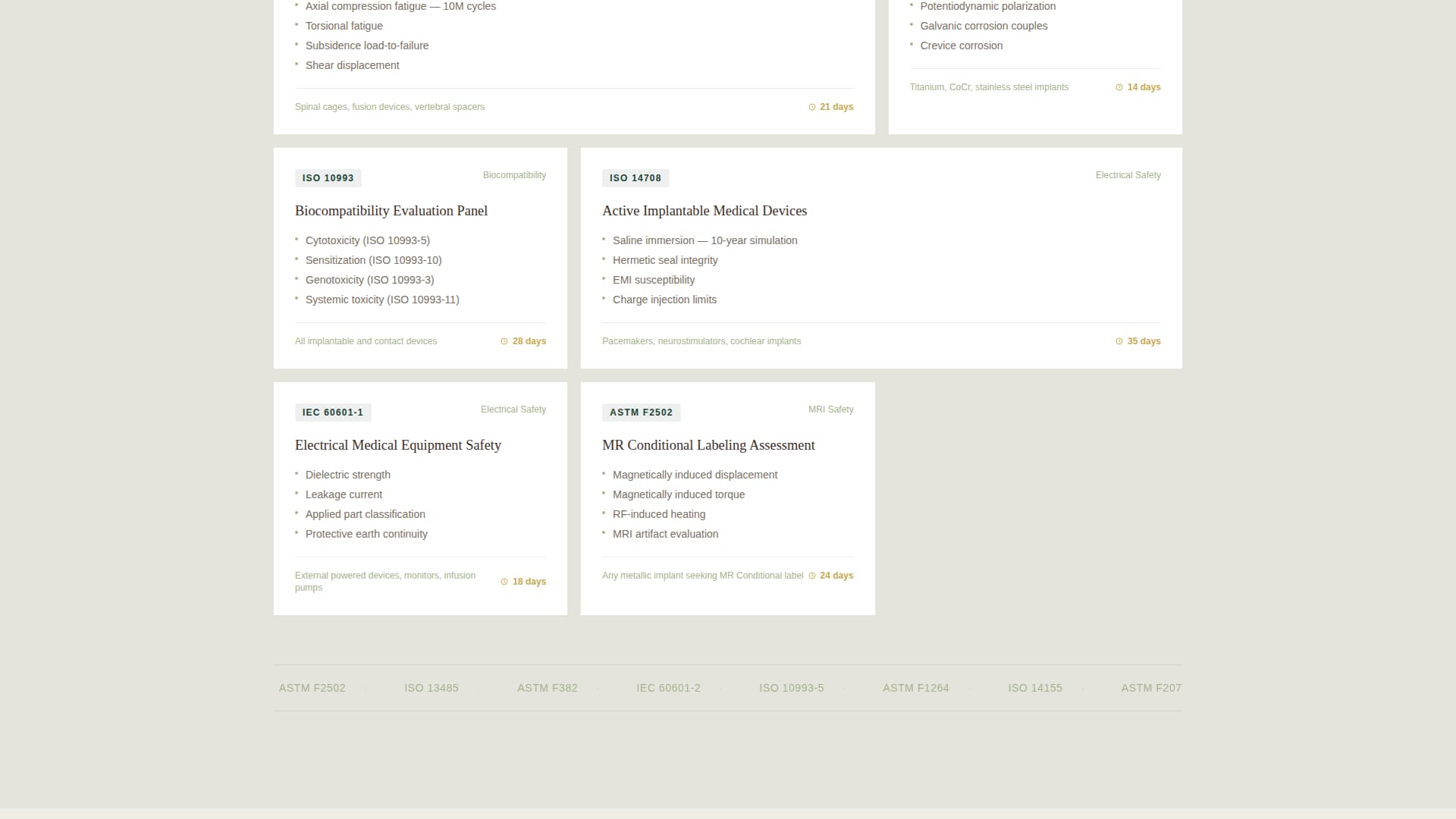
Capability Matrix by Test Standard
The template includes a structured capability matrix organized around recognized test standards such as ASTM F2077, ISO 14708, and IEC 60601. This layout helps technically trained visitors quickly locate the protocols most relevant to their device classification without hunting through dense prose.
Ungated Data Tables and Infographic
Two complete data tables and a regulatory-timeline infographic are embedded in the page without a gate. This approach demonstrates the lab's systems thinking before requesting any contact information, lowering visitor skepticism and improving conversion quality.
Gated Testing Roadmap Download Form
The primary conversion element is a form that offers a PDF guide mapping device classification to required test protocols. The form collects work email, company name, and device type via a dropdown that includes implantable, external, in vitro diagnostic, and combination device categories.
Persistent Secondary Call-to-Action Button
After the third scroll section, a gold-outlined "Request a Test Plan Review" button appears in the lower right of the viewport. It remains accessible as visitors continue scrolling without interrupting the reading experience, creating a second conversion path for buyers further along in their decision process.
Page sections overview
| Section | Purpose |
|---|---|
| Video Header | Establishes lab credibility and surfaces the primary headline |
| Market Context Paragraph | Anchors the problem with FDA warning-letter statistics |
| Capability Matrix | Organizes test standards by protocol for quick scanning |
| Ungated Data Table One | Delivers immediate value with pass/fail outcomes and cycle counts |
| Regulatory Timeline Infographic | Visualizes turnaround benchmarks ungated |
| Ungated Data Table Two | Reinforces evidence with a second set of anonymized case-study data |
| Accreditation Logo Strip | Displays accreditation marks alongside turnaround benchmarks |
| Gated Download Form | Captures qualified leads with the Testing Roadmap PDF offer |
| Persistent call to action Button | Surfaces test plan review path from the third section onward |
Design & branding system
The visual identity follows a Corporate Precision theme using a Forest Trust color system. The palette is inspired by a Pacific Northwest forest at dawn, composed and deeply trustworthy in character.
- Core colors are old-growth evergreen (#1B4332), lichen gray (#A3B18A), birch-bark white (#F0EDE5), and deep loam (#3A2D1F) for body text, with a quiet gold (#C9A84C) reserved for download icons and hover states
- Backgrounds alternate between birch-bark white and a faint five-percent evergreen wash; section dividers are thin lichen-gray rules that breathe rather than divide
- Typography and layout follow an industry-report cadence, with each section visually distinct through spacing and divider lines rather than heavy color blocking
Mobile & speed optimization
The single-column flow structure ensures the page reads cleanly on any screen width. The linear content sequence translates directly to a mobile scroll without requiring layout changes.
- Single-column layout means content reflows naturally from desktop to tablet to mobile without horizontal overflow or complex grid adjustments
- The persistent secondary call-to-action button is positioned to remain accessible on smaller screens without covering primary content
- Section dividers and alternating backgrounds maintain visual separation on small screens without relying on multi-column grids
How this template helps you convert
The conversion strategy is built around earning trust before requesting information. The page sequences content so that visitors accumulate evidence before they encounter any form.
- Ungated tables and the regulatory-timeline infographic are placed early in the scroll so visitors receive concrete value before seeing the download form, which meaningfully reduces resistance at the gate
- The gated Testing Roadmap form uses a focused four-field structure collecting work email, company name, and device type, keeping the ask proportionate to the value offered
- The persistent gold-outlined secondary button creates a parallel conversion path for visitors who are ready to discuss a specific test plan, capturing buyers at two different stages of readiness
Other information about this template
This template is suited to manufacturing and industrial contexts, particularly within the medical device manufacturing space. It is built to serve the medical device quality testing lab niche where credibility and precision are the core marketing currency.
- The template style is a single-column flow, making it straightforward to customize section content and swap in real lab footage, data tables, and accreditation logos
- The creative direction follows an industry-report cadence, making it compatible with content strategies that prioritize technical depth over promotional tone
- The Forest Trust color system and Corporate Precision theme can be adapted by adjusting hex values while keeping the overall visual rhythm intact
Theme
Corporate Precision
Creative direction
Industry Report
Color system
Forest Trust
Style
Single Column Flow
Direction
Content/Resource
Page Sections
Full-screen Video Background Header
Industry Report Content Cadence
Test Standard Capability Matrix
Ungated Tables and Infographic
Gated Testing Roadmap Download Form
Persistent Secondary Conversion Button
Related questions
Can I replace the video background with my own lab footage?
What device types does the dropdown in the download form support?
Is the capability matrix content editable?
When does the persistent secondary call-to-action button appear?
Does the template include the PDF guide or real data table content?